Healthcare Professionals
Allegra has no brain interference and no sedative effect1
For patients with tough allergy symptoms, that's a no-brainer
Every patient is different and can have a variety of symptoms.* Allegra provides powerful allergy relief for patients with itchy, watery eyes, itchy throat, runny nose, and sneezing.† It starts working in 1 hour and doesn't cause drowsiness. That's why it’s the #1 allergist-recommended non-drowsy OTC antihistamine brand.‡

Learn more about:
No Brain Interference from Drowsiness
Allegra has no brain interference and no sedative effect 1
- Fexofenadine, the antihistamine molecule in Allegra is proven in several controlled studies to have 0% brain interference
- Any brand that has less than 20% brain occupancy can claim to be non-drowsy
- Brands like Zyrtec can cause drowsiness which can impact cognitive performance
Brain histamine H1-receptor occupancies of various antihistamines and classification for sedating actions. The occupancy data are represented as the mean +/- SD of measurements in positron emission tomography after oral single-dose administration of the drugs
.4187473839216389154.870412512397475771.2025-02-14-13-51-25.2025-10-21-09-23-03.webp)

Efficacy
Allegra acts fast
One hundred forty-six subjects who adequately responded to priming entered a single-dose placebo phase. Placebo-responders were disqualified from the study, leaving other subjects with adequate symptoms to be randomized and given a single dose of either fexofenadine HCl 120 mg, 60 mg or placebo, after 60 minutes of allergen exposure.
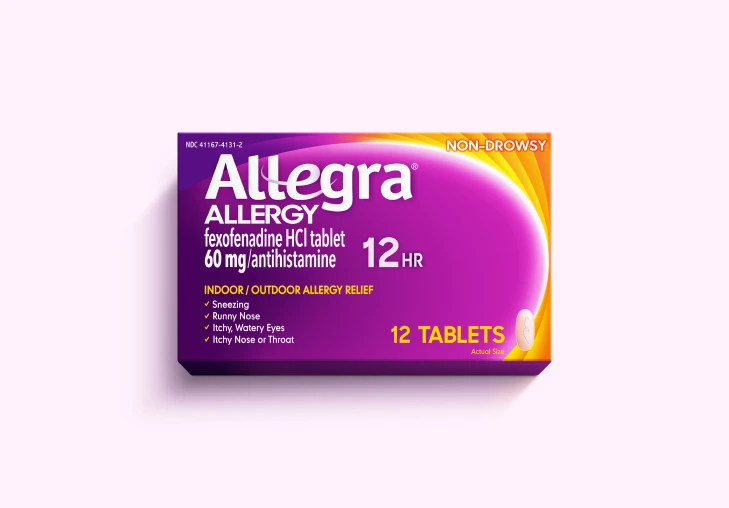
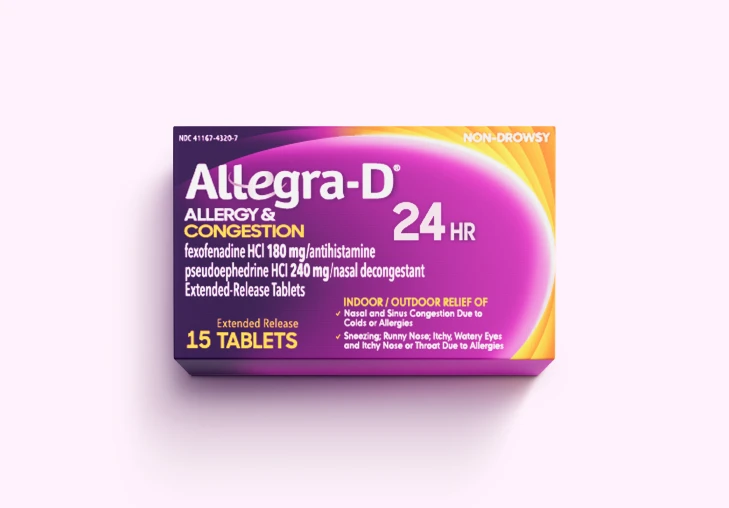
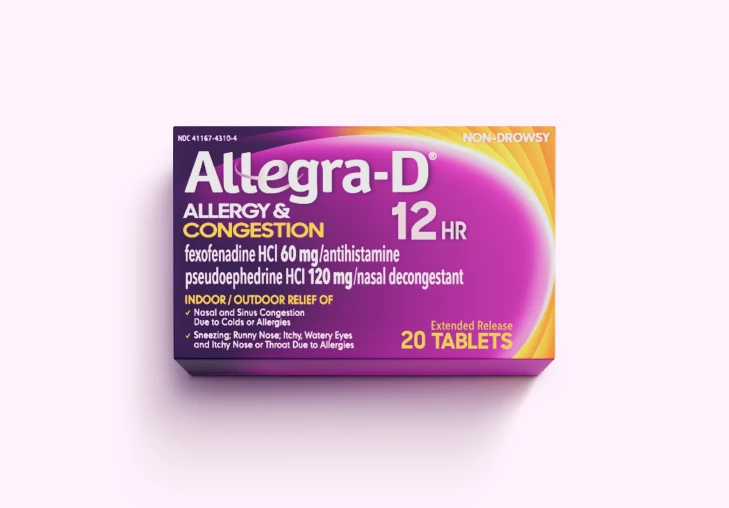
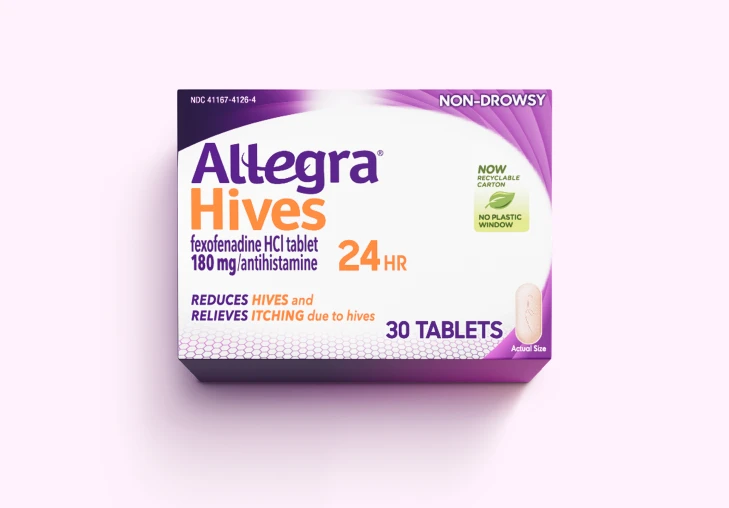
Allegra® Products
Get relief from the #1 allergist-recommended non-drowsy OTC antihistamine brand‡ for tough allergy symptoms* with Allegra® Allergy, Allegra-D®, and Children's Allegra® products.
Allergy Relief
Allergy & Congestion Relief
Hives Relief
Relief for Kids
FAQs
Does Allegra-D® contain phenylephrine (PE)?
No. Allegra-D® does not include phenylephrine. It's formulated with pseudoephedrine (PSE), a clinically proven ingredient for nasal congestion relief.
Does Allegra® Allergy 24-hour tablets contain phenylephrine (PE)?
No, Allegra® Allergy 24-hour tablets do not contain phenylephrine (PE). The active ingredient in these products is fexofenadine, a non-drowsy antihistamine. Fexofenadine effectively relieves allergy symptoms such as sneezing, runny nose, itchy/watery eyes, and an itchy nose or throat. These symptoms can be caused by hay fever or other upper respiratory allergies.
Does Children's Allegra® Allergy contain phenylephrine (PE)?
No, Children's Allegra® Allergy relief does not have phenylephrine in its formulation. Children's Allegra® is formulated with Fexofenadine HCl, a non-drowsy antihistamine that works to relieve allergy symptoms of sneezing, runny nose, itchy/watery eyes, and itchy nose or throat due to hay fever or other upper respiratory allergies.
Does Allegra® Hives contain phenylephrine (PE)?
No, Allegra® Hives does not have phenylephrine in its formulation. It is made with Fexofenadine HCl, a non-drowsy antihistamine that reduces hives and relieves the itching caused by hives.
References
1. Ansotegui IJ, Bousquet J, Canonica GW, et al. Why fexofenadine is considered as a truly non-sedating antihistamine with no brain penetration: a systematic review. Curr Med Res Opin. 2024 Aug;40(8):1297-1309.
*Symptoms include itchy, watery eyes; itchy nose or throat; rhinorrhea; and sneezing.
†See Drug Facts label. Use as directed.
‡Among single-ingredient branded OTC oral antihistamines.